|
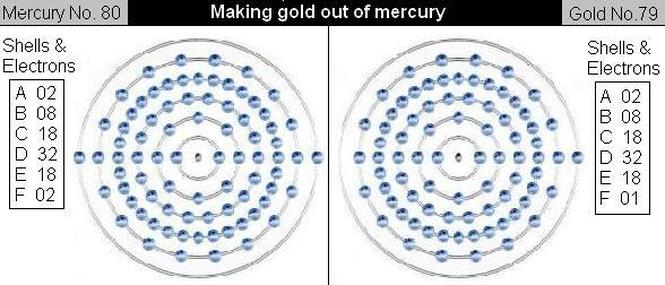
By G. Hunter With precious metal prices constantly on the rise, I've been exploring the possibility of making some of my own gold. Making gold is not as difficult as you would imagine, all you need is one alchemist with specific esoteric knowledge or, more realistically, a nuclear reactor capable of nuclear transmutation. Transmutation of the elements has been explored by men and women a lot longer that you think, while modern methods of transmutation have become simpler due to technological innovations - innovations that are often misleading as to what their true capabilities are. Video Description: In March of 1924 Man Discovered the Secret of Alchemy. Since 1954, 31 countries have built 435 nuclear power plants. I'd say all of them were designed to preform this operation. Generating electricity may not be the primary use, as we have been told. They have had the ability to commercially produce gold for 58 years. 24 hours a day, 7 days a week. There really is no telling how much they have stock piled.. Mercury's current market price = $1.25 oz. / Gold is $1,655 oz. In March 1924, at the Tokyo Imperial University, Professor Hantaro Nagaoka directed 150,000 volts of electricity at a mercury isotope under a dialectic layer of paraffin oil for four hours in an early experiment with nuclear energy. The purpose was to strike out a hydrogen proton from the nucleus of the mercury and produce a new element, gold. Mercury has 80 protons. Gold, meanwhile, has 79 protons — you see where I’m going with this.The experiment was a success. Professor Hantaro Nagaoka solved the mystery that eluded scientists for centuries, the mystery of the Philosopher’s Stone. The Philosopher’s Stone is the idea that you could have a magical material that could turn lead, or some very inexpensive metal, into gold. For thousands of years, kings sought out this mythical device, one that could create gold out of common metals. Scientists and alchemists for centuries have been trying to invent one. Even Sir Isaac Newton obsessed over the mystery of the Philosopher’s Stone in the 17th century. However, the English feared the potential devaluation of gold and made the practice of alchemy punishable by death. Fast forward now a few centuries to present day. If we wish to manufacture gold, the most helpful metal to start with is mercury. Gold is element 79 and mercury is element 80, which means that there is only a slight difference between their atomic structures. The mercury atom has one more proton in its nucleus and the corresponding electron in the outer (known as F shell) orbit. As the diagram shows, all other shells (from atom A to E) have the same number of electrons in both mercury and gold. So, theoretically, if we can expel one proton from the nucleus of an atom of mercury, we have transmuted it into an atom of gold. The process is difficult since an atom of mercury has eighty electrons; eighty orbits have to be broken through as well as the electric field round the nucleus. The first experiment was, however, carried out years ago at the Physical-Technical State Institute of Berlin. The bombarding particles were given a high speed by means of a field of 30,000 volts, and a small, but observable quantity of gold was produced from quicksilver. Unfortunately, such laboratory transmutation can never be reproduced on a commercial scale.
It is tempting to laugh off medieval alchemists as greedy eccentrics, who sought methods for forming gold out of cheaper metals. But one ought to give them credit for what they did in the process of searching. These alchemists discovered strong acids like hydrochloric acid, nitric acid and sulfuric acid which are far more useful today then gold could possibly be. The alchemists should have been acclaimed for these revolutionary discoveries. Instead they were sneered at for their failure to make gold out of plentiful metals like mercury. Before Chemistry was a science, there was Alchemy. One of the supreme quests of alchemy is to transmute lead into gold. Lead (atomic number 82) and gold (atomic number 79) are defined as elements by the number of protons they possess. Changing the element requires changing the atomic (proton) number. The number of protons cannot be altered by any chemical means. However, physics may be used to add or remove protons and thereby change one element into another. Because lead is stable, forcing it to release three protons requires a vast input of energy, such that the cost of transmuting it greatly surpasses the value of the resulting gold. How to make Gold from Mercury
What about Lead? Transmutation of lead into gold isn't just theoretically possible - it has been achieved as well. There are reports that Glenn Seaborg, 1951 Nobel Laureate in Chemistry, succeeded in transmuting a minute quantity of lead (possibly en route from bismuth, in 1980) into gold. There is an earlier report (1972) in which Soviet physicists at a nuclear research facility near Lake Baikal in Siberia accidentally discovered a reaction for turning lead into gold when they found the lead shielding of an experimental reactor had changed to gold. Today particle accelerators routinely transmute elements. A charged particle is accelerated using electrical and/or magnetic fields. In a linear accelerator, the charged particles drift through a series of charged tubes separated by gaps. Every time the particle emerges between gaps, it is accelerated by the potential difference between adjacent segments. In a circular accelerator, magnetic fields accelerate particles moving in circular paths. In either case, the accelerated particle impacts a target material, potentially knocking free protons or neutrons and making a new element or isotope. Nuclear reactors also may used for creating elements, although the conditions are less controlled. In nature, new elements are created by adding protons and neutrons to hydrogen atoms within the nuclear reactor of a star, producing increasingly heavier elements, up to iron (atomic number 26). This process is called nucleosynthesis. Elements heavier than iron are formed in the stellar explosion of a supernova. In a supernova gold may be made into lead, but not the other way around. While it may never be commonplace to transmute lead into gold, it is practical to obtain gold from lead ores. The minerals galena (lead sulfide, PbS), cerussite (lead carbonate, PbCO3), and anglesite (lead sulfate, PbSO4) often contain zinc, gold, silver, and other metals. Once the ore has been pulverized, chemical techniques are sufficient to separate the gold from the lead. The result is almost alchemy...almost.
7 Comments
PETER
6/24/2014 06:43:09 pm
IM SURE THEY DO, WHY ELSE WOULD THEY ALLOW CHINA, RUSSIA AND INDIA BUY SO MUCH GOLD. THEY MAKE IT AND SELL IT AND HOLD ON TO THE SECRET. THERE IS NO NEED TO KNOW ESPECIALY IN THIS MATTER.
Reply
Bob
6/24/2014 07:10:15 pm
I had a physics professor in college who turned lead into gold using a particle accelerator. It wasn't economically viable because of the amount of energy used.
Reply
vk
3/18/2017 08:16:43 am
It is not that tough, if you know the exact procedure. There are so many indian methods to make Gold from Mercury....My Grand father told me so many times about this, when i was in 10th grade
Reply
tw
6/25/2014 03:02:05 am
so gold is becoming worthless
Reply
BTCMAN
5/21/2017 05:14:58 am
This is why Bitcoin has all the more reason to continue gaining the traction it's been taking. Never too late to get in on this one. With BTC, there's an absolute finite amount in circulation. Take that to the bank!
Reply
Eric S. Harris
4/18/2022 01:44:02 am
Nope. Before the electrolytic process for refining aluminum was discovered, aluminum was more expensive that gold.
Reply
Whocouldshebe
8/27/2017 10:25:23 am
Neon sign transformers generate extremely high voltage, the plan I came up with was to bombard the Mercury with 20000 volts covered with a layer of paraffin wax. Wouldn't that be a cheap alternative to avoid $5000 an hour for a particle accelerator?
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
News Watch
Mind-opening news articles, editorials, videos & apparel that inspire our readers and help liberate them from the status quo. Stay informed.
Write For UsSpace WatchTop NewsNews Watch Categories
All
|
|
|
HAVE A TIP OR STORY TO TELL? JOIN TODAY & SHARE YOUR STORY!
If you have a breaking news tip or idea, please email: tips@apparentlyapparel.com Apparently Apparel® is a registered trade name and part of the ZOAT International® brands network. © 2007-2023. All Rights Reserved. Privacy Policy. All art & news content posted on this site is commentary or opinion and is protected under Free Speech. ApparentlyApparel.com is not responsible for content written by contributing artists, authors or news feeds. The information on this site is provided for educational and entertainment purposes only. It is not intended as a substitute for professional advice of any kind. ApparentlyApparel.com assumes no responsibility for the use or misuse of this material.
|
|